Daily arthritis pill could slow progression of diabetes, trial finds

A daily prescribed daily pill for rheumatoid arthritis and alopecia can also slow down the progression of type 1 diabetes.
The chronic disease caused by the immune system that accidentally attacks insulin -producing cells in the pancreas affects approximately 270,000 people living in the UK and many of them have to take life -long insulin injections to help manage blood sugar levels.
However, a two -year Australian study found that the drug Barisitinib, who works by blocking signals leading to over activity of the immune system, helped to protect the remaining insulin -producing cells in newly diagnosed people.
Researchers believe that this drug, which is used to treat several autoimmune disease, including rheumatoid arthritis, ulcerative colitis and alopecia, can delay the progression of type 1 diabetes symptoms.
Dr. Australia, Fitzroy’dan ST Vincent’ın Medical Research Institute. Michalea Waibel said: “For the first time, we have a modifying treatment that can intervene early enough to allow people with type 1 diabetes to be significantly dependent on insulin therapy, and provide time -free time from the demands of the daily management of the disease and provide lower proportions of long -term complication rates.
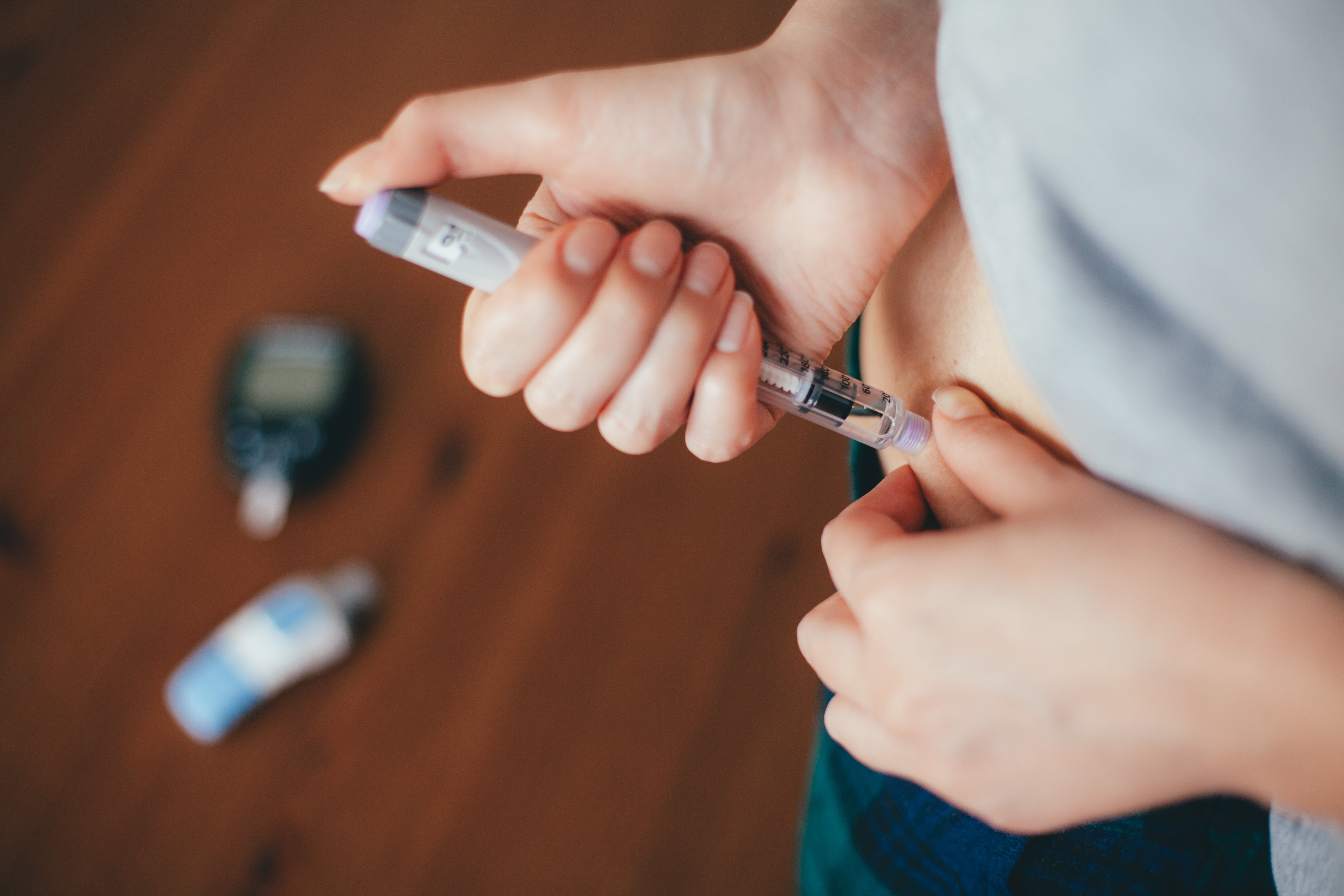
The first trial in 2023 has recently enrolled in 91 people who have recently diagnosed Type 1 diabetes in the last 100 days. The participants were given a barisitinib pill (4mg) or a placebo once a day for 48 weeks.
Participants C -peptide levels – a marker of insulin secretion – followed the glucose monitoring and HBA1Cs to find out how much participants can do themselves and also control blood sugar levels.
Researchers have found that the protection of baricidinibine found insulin -producing cells reduce blood sugar fluctuations and reduce the need for insulin.
Now the results of a follow -up hearing have been announced. European Diabetes Study Association After the drug was stopped, the diabetes of the participants progressed.
Researchers found that the C-peptide level in 48 weeks was 0.65 in the Barisitinib group and 0.43 in the placebo group. After the treatment was stopped, the C-peptide levels decreased to 0.49 NMOL per liter in the barisitinib group and to 0.36 in the placebo group in 72 weeks and then to 0.37 and 0.26 in 96 weeks and insulin production decreased.
When Barisitinib treatment was stopped, the participants needed more insulin treatment with requirements that were not significantly different from the placebo group in 72 and 96th weeks.
During the follow -up period, additional security concerns did not emerge, and researchers hope that the drug may be available for diabetes in the next five years.
“If we can define people with high risk of developing type 1 diabetes with genetic tests and blood markers, they can be treated earlier to prevent the disease from being kept in the first place, Dr Dr. Waibel added.
“We hope that the larger phase III studies with Barisitinib will soon begin to delay insulin addiction in recently identified types of diabetes and previous stages.”




